
A guide to Oblique Contrast
Oblique contrast is a microscopy technique used to increase the contrast of images of thin, transparent, unstained samples, the details of which can be difficult to visualise using brightfield microscopy. Two main advantages of oblique contrast are that it is a cheap and easy way of improving the contrast of your images.
Similarly to Differential Interference Contrast (DIC), images produced using oblique contrast have shadowed regions, giving a pseudo three-dimensional effect. The light strikes the specimen at an oblique angle, rather than an axial angle, which introduces phase changes to reveal features with a much greater contrast than brightfield illumination.
Applications
Oblique contrast is suitable for imaging thin, transparent or nearly transparent samples such as cell cultures or thin brain slices. It can be used for visualising specimens that can’t be stained, such as living cells, tissues or organisms.
As oblique contrast doesn’t use polarised light, it is suitable for imaging vertebrate axons. Vertebrate axons are myelinated by a birefringent material and, unlike polarised light, the unpolarised light doesn’t affect the refractive index of the myelin. As petri dishes are also birefringent, oblique contrast is ideal for imaging samples within these, for example cultures of bacteria.
There are no additional optics in the above sample collection path, making oblique contrast ideal for two-photon and confocal microscopy, as no intensity is lost. It is also used to introduce contrast in patch-clamp experiments.
Advantages
- As oblique contrast does not use polarised light, it is not affected by birefringent samples so is ideal for imaging these. (Birefringent samples have a different reflective index depending on how the light is polarised).
- It is simple to use – a small disc is moved around to adjust the image.
- It is cheaper than other methods, such as DIC (Differential Interference Contrast).
- It produces pseudo three-dimensional images to aid techniques such as patch-clamping.
Limitations
- Oblique contrast can generate false images in some samples, depending on the angle that the light goes through the sample.
- The same specimen can appear very different if the orientation of it is changed which can be misleading.
- The 3D effect is not representative of the actual geometry of the specimen, so you can see 3D depths that aren’t real, similar to optical illusions.
- Oblique contrast may affect image resolution as the full numerical aperture of the objective and condenser is not being utilised.
How it works
With a standard condenser used in brightfield microscopy, the specimen is axially illuminated; light passes through the centre of the specimen, diffracting at different angles as it hits the specimen before recombining at the back of the image plane. As the light diffracts at different angles and therefore travels a longer distance before reaching the image plane, the phase of the light is affected. However, when light of equal amplitude and wavelength and inverse phase combines it cancels out so there is no overall change in phase. This is called destructive interference. Therefore, there is no overall change in the image.
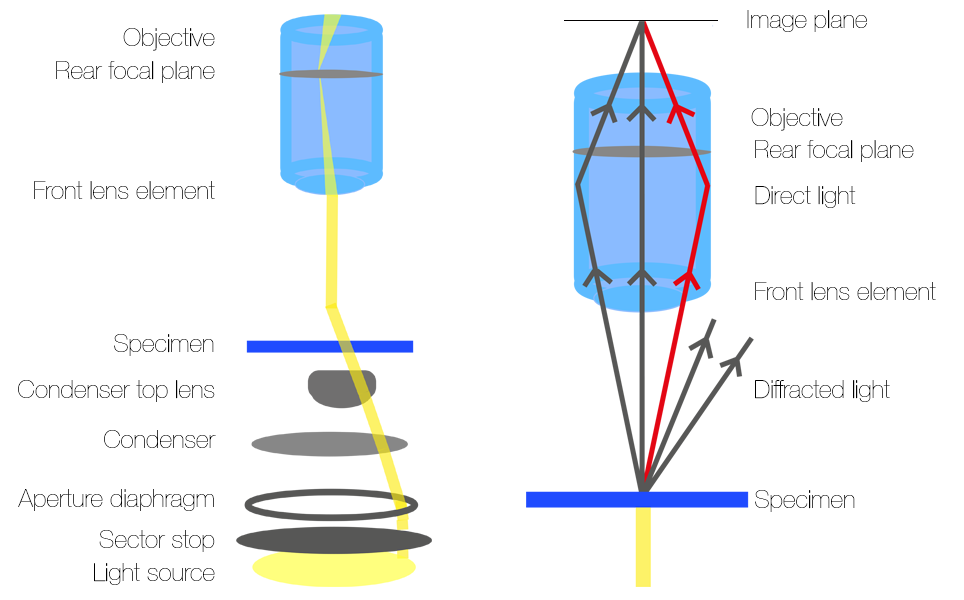
Oblique contrast utilises an oblique condenser containing a sector stop with a small off-centre slit which makes the light go through the sample at an angle, so the specimen is illuminated from one direction only, shown in figure 2a. The sector stop is located beneath the lens and aperture diaphragm of the condenser. Light from the source passes through the slit which causes it to pass through the condenser lens at an oblique, off-axis angle. The specimen is then illuminated an angle.
After passing through the specimen, the light rays enter the microscope objective and converge at the rear focal plane before they are observed through eye pieces or on a camera. As the light in oblique contrast hits the sample at an angle, only the diffracted sidebands of light on one half of the direct beam will be collected by the objective, as shown in figure 2b. This means that there is constructive and destructive interference between the direct light beams and collected sidebands.
Set-up
To make a brightfield microscope capable of oblique contrast, the only additional components needed are an oblique condenser and an infrared or visible light source. The condenser is placed in the condenser housing above the light source. The oblique contrast mechanism (sector stop) of the condenser is located below the condenser lens and aperture diaphragm.

Firstly, pull out the oblique slider and open the oblique iris so the condenser is adjusted for brightfield illumination.

Set up Koehler illumination on the microscope. If using a manual microscope, remove the condenser stop before setting up Koehler illumination and replace it once Koehler illumination has been set up.
Read Scientifica’s Koehler illumination guide
Close down the condenser iris to match the numerical aperture of the objective.

Then, adjust the direction of the sector stop by turning the diaphragm control knob. Close down or open the stop to adjust the amount of oblique illumination until the desired image is achieved. The amount of oblique illumination needed depends on the specimen and the amount of detail required. The oblique condenser enables the angles of illumination to be altered through 360 degrees without moving the specimen.

When making adjustments, check that the sector stop is still positioned correctly and that the slider is not too far in or out.
Your microscope is now set up for oblique contrast. Please contact Scientifica if you have any questions or require any further guidance.
We have created a downloadable PDF of this guide, download it here:
Oblique Contrast
Learn about other contrast techniques
Sign up to receive our latest news
Find out about Scientifica's latest product releases, company news, and developments through a range of news articles, customer interviews and product demonstration videos


