Using a Scientifica Multiphoton Microscope to study neurophysiology in the CNS
Two-photon microscopy is an important technique in the study of neurophysiology. It enables visualisation of the functional and morphological properties of neurons and their networks deep into living tissue with high spatiotemporal resolution. This makes it an elegant tool for studying the basic physiology of the central nervous system either in vivo or in vitro.
Scientifica’s multiphoton systems have been used for a broad range of experiments on the CNS and can be easily combined with other techniques, such as electrophysiology or optogenetics, to help researchers uncover the secrets of the brain. This article introduces four examples of experiments where the use of a two-photon microscope has enabled this.
Linking synaptic activity to calcium signalling: simultaneous electrophysiology and two-photon calcium imaging
Neurons receive thousands of synaptic inputs which if activated synchronously in sufficient numbers cause action potentials. But synapses are not static relay points for the electrical signals travelling within neural circuits; they are also a key component of the brain’s plasticity mechanisms allowing adaptation of behaviour to changes in the environment.
Understanding how synaptic activity encodes the magnitude and direction of synaptic plasticity can uniquely be achieved by integrating electrophysiology with two-photon fluorescence imaging of spine Ca2+ transients.
Learn more
Astrocytes respond to inhibitory neurotransmitter GABA
Researchers from the University of Padova in Italy have discovered that cortical astrocytes respond to the inhibitory neurotransmitter GABA through GABAB receptor-mediated calcium oscillations in developing and adult mice.
Through confocal and two-photon laser scanning microscopy of the somatosensory and temporal cortex, the researchers show that, in vitro, the application of GABA or Baclofen (a GABA agonist) evoked Ca2+ oscillations in the soma of a subpopulation of astrocytes. Further in vivo experiments also exhibited GABA receptor mediated calcium elevations.
Lose your inhibitions
Ion channel function in the absence of ion-flow
Dr Roberto Malinow’s lab at the University of California, San Diego, shows that agonist binding to the NMDA receptor (NMDAR) triggers conformational movement of the cytoplasmic domain (NMDARcd) in the absence of ion flow.
The researchers used two-photon fluorescence lifetime imaging microscopy (FLIM) to measure Förster Resonance Energy Transfer (FRET) between GluN1-GFP and GluN1-mCherry. FRET between the fluorescently tagged cytoplasmic domains of the GluN1 subunits of NMDARs is reduced rapidly and transiently with agonist binding. These results are consistent with a conformational change on the NMDARcd.
Go with the flow
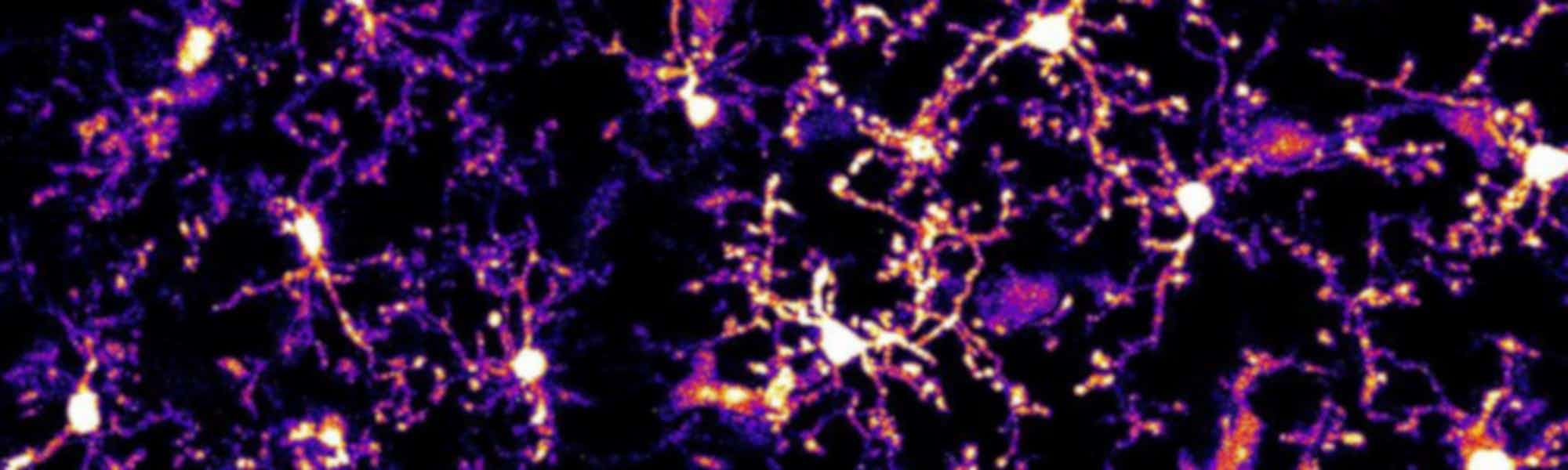
Neuron-microglia communication in the CNS after epileptic seizures
The way that neurons regulate microglia is not well understood. However, recent research has illuminated the mechanism by which neurons control morphological changes in microglia after induced seizure activity.
Seizures are associated with a significant increase in extracellular glutamate, causing a spike in neuronal activity. These periods of intense neural activity also lead to alterations in the shape of microglia, as visualised two-photon time-lapse microscopy.

)