
The most exciting neuroscience research from May 2021
Scientifica's selection of the most exciting neuroscience research from May includes discovery of the normal physiological function of the APP protein family, a brain-computer interface that could enable paralysed people to communicate and a promising development of a nasal spray treatment for Parkinson’s disease.
1. Composing thoughts: mental handwriting produces brain activity that can be turned into text
Scientists at HHMI, funded by the NIH BRAIN Initiative, have developed a brain-computer interface (BCI) that could restore the ability to communicate in people with spinal cord injuries and neurological disorders. The BCI works by translating brain activity related to intended handwriting into text.
In the research, brain signals generated when a person, who was paralysed from the neck down due to a spinal cord injury, attempted to write letters by hand were recorded, and a machine learning algorithm was trained to identify neural patterns that represent individual letters. In this proof-of-concept study, the system appeared to be more accurate and efficient than existing BCIs, and has the potential to enable people with paralysis to rapidly type without using their hands.
The technology also has the potential to enable those living with ‘locked-in syndrome’ caused by brainstem stroke or late-stage ALS, who are unable to write or speak, to communicate again.
Translating handwriting in the brain
2. Mini 3D brain models to speed MS research
Tiny 3D models that mimic aspects of the human nervous system have been developed by scientists at the University of Edinburgh.
The models are the most natural representation of human myelination that have been developed in a lab, and could therefore enhance research into neurological diseases that are linked to myelin loss, such as multiple sclerosis.
The human myelin model was developed using skin samples donated by volunteers. The skin cells were reprogrammed into induced pluripotent stem cells, which were then developed into spinal cord cells. Organoids, 3D structures of cell bundles, were then grown from these cells.
In a model developed from a healthy donor, the myelin developed and functioned as it would in a healthy brain or spinal cord. However, in an organoid created using stem cells from a patient with a gene mutation that affects myelination, there were some key features that matched the disease.
The new models will enable researchers to compare the differences between the cells of healthy individuals and those with different neurological conditions, as well as test drugs on human cells before the full clinical trial stage.
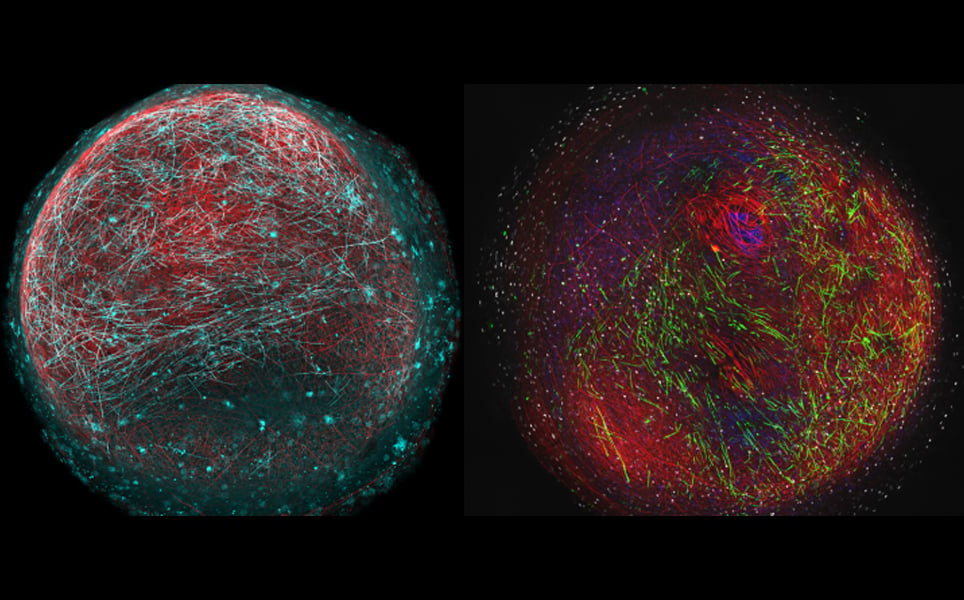
Mimicking myelination
3. Cell-type parts list of the mouse neocortex and the hippocampal formation yields new information about how the mammalian brain evolved
A study by the Allen Institute has mapped all the types of neurons, as well as many other types of cells, in the adult mouse neocortex and hippocampal formation.
These two structures are essential for the cognitive function of the mammalian brain. Mapping these cells showed that the hippocampal formation, one of the older parts of the brain, is just as complex as the neocortex, a more recently evolved area of the brain.
Using single-cell transcriptomics, a dataset of more than 1.3 million cells was produced, which has led to new insights into evolution and the development of the mammalian brain.
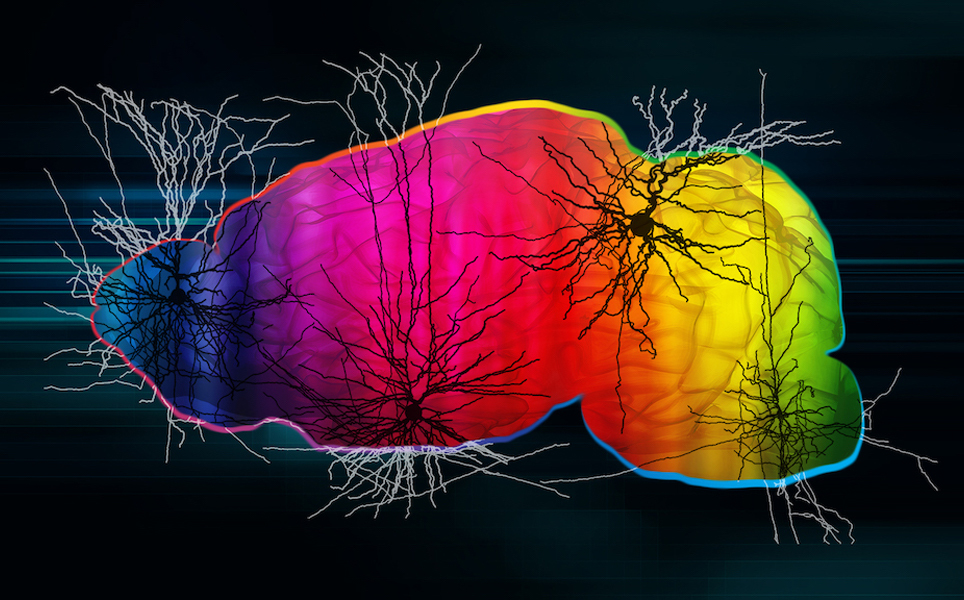
Learn more here
4. Beyond plaques: Unravelling the natural functions of the APP protein family
An international team of researchers led by Heidelberg University has discovered the role of the APP protein family in the healthy brain.
Although the APP protein is known to be involved in Alzheimer’s disease, until now, its role in healthy brain function was unknown. The research showed that a mouse model lacking APP during brain development had malformations in the layered structure of the hippocampus. As this brain region is essential for learning and memory, this resulted in the mice having impaired learning abilities, with the mice displaying behavioural changes resembling those seen in autism spectrum disorder.
The findings suggest that the APP protein family plays an essential role in the normal development of the nervous system, learning and memory and social communication. As APP is produced by most cells in the brain, understanding the normal physiological function of this protein may help with the development of novel therapeutics for Alzheimer’s disease.
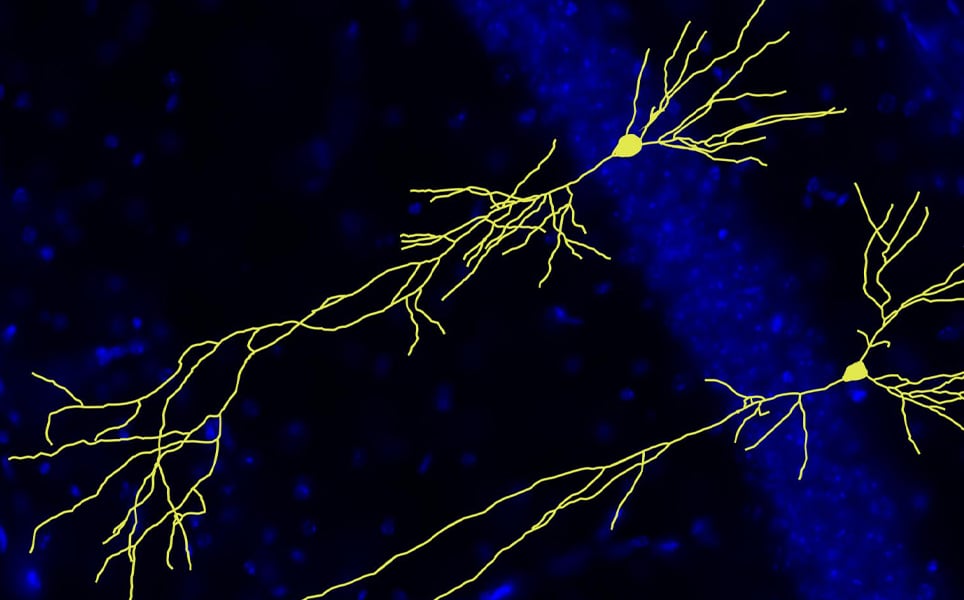
Understanding APP
5. How injured nerves stop themselves from healing
A discovery made by researchers at Ruhr-Universität Bochum (RUB) could explain why damaged nerve fibres in the central nervous system are unable to regenerate, which could lead to opportunities for the development of new treatments.
Results suggest that damaged nerves release a protein which attracts growing nerve fibres, trapping them at the site of injury. The growing nerve fibres are then unable to continue growing in the right direction, in order to repair the injury.
The protein, a chemokine called CXCL12, is released at the site of a nerve lesion. This protein is so chemoattractive that growing nerve fibres become trapped, with some fibres that had already regenerated across the injury site changing direction, and growing back towards the injury.
As this protein appears to be a major inhibitor of neural repair, it is a new therapeutic target. The researchers found that knocking out CXCL12 in retinal nerve cells, so it could not be released at the injury site, improved axonal regeneration.

Potential for improving nerve repair
6. Robotic ‘Third Thumb’ use can alter brain representation of the hand
A study led by researchers at UCL has shown that using a robotic ‘Third Thumb’ can change how the hand is represented in the brain.
After training people to use the robotic extra thumb, the team found they could carry out dextrous tasks, such as building a tower of blocks, with only one hand. Participants also increasingly felt like the extra thumb was part of their body.
The device was first developed by designer Dani Clode, with the aim of reframing the way we view prosthetics, from replacing a lost function, to an extension of the human body. She then joined Professor Tamar Makin’s team at UCL, who were investigating how the brain can adapt to body augmentation.
As the Third Thumb is 3D-printed, it is easily customised, and is worn on the opposite side of the hand to the user’s real thumb. It is controlled using pressure sensors on the underside of the big toes of the user’s feet, which are connected wirelessly to the thumb.
20 participants were trained to use the thumb over five days, and they were able to quickly learn to control the device, changing their natural hand movements while using the Third Thumb. Subtle changes in the brain’s sensorimotor cortex were detected in fMRI scans, which showed that the brain activity pattern corresponding to each individual finger had become less distinct in users of the Third Thumb.

The future of prosthetics?
7. Gene therapy restores partial vision to a blind patient
Scientists at the University of Basel and University of Pittsburgh have successfully used optogenetics to restore partial vision to a patient who had become blind due to the inherited disease retinitis pigmentosa.
Optogenetics was used to restore the light-sensing capability of the patient’s retina. The researchers delivered genes to the retina’s ganglion cells which stimulated them to produce ChrimsonR, a light-sensitive protein which senses amber light; a safer light for retinal cells than the blue light which is used for other types of optogenetics. Specialised goggles were also developed, containing a camera that captures and projects images onto the retina at amber light wavelengths.
The patient began reporting signs of visual improvement seven months following the gene therapy, and was able to locate, touch and count objects placed on a white table in front of him, only whilst wearing the goggles. EEG recordings of brain activity whilst performing a task showed that the activity of the visual cortex had changed according to whether an object was present or not, showing that brain activity related to a visual object, and the retina was no longer blind.
This treatment is hoped to be a viable option for patients with a functional optic nerve who have lost their vision due to a neurodegenerative condition that causes photoreceptor degradation.

Breakthrough in vision restoration
8. NIH study identifies diverse spectrum of neurons that govern movement
A diverse range of motor neurons throughout the spinal cord, including a subset that is susceptible to neurodegenerative diseases, have been identified and mapped by researchers at the National Institutes of Health.
Traditionally, spinal cord neurons have been categorised into three main types: skeletal motor neurons, visceral motor neurons, and interneurons. This atlas, created using single nucleus RNA sequencing, has identified 21 subtypes of neurons in discrete areas along the spinal cord. The team found that motor neurons that control glands and internal organs have particularly distinct subtypes, and that visceral motor neurons extend higher up the spinal column than previously thought.
This research provides new insight into how movement throughout the body is controlled, and why some neurons are more susceptible to neurodegenerative diseases such as spinal muscular atrophy and amyotrophic lateral sclerosis.
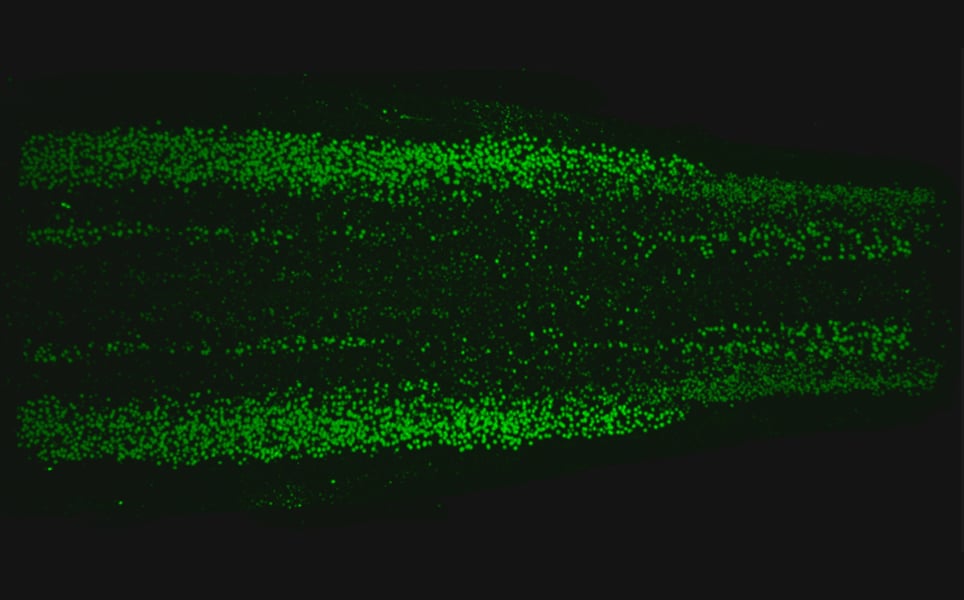
Learn more
9. Aggressive brain tumours can mimic normal brain repair processes
Researchers at UCL have discovered that glioblastoma tumour cells mimic brain repair; when using white matter to spread to other brain regions, they attempt to repair the damage caused by the growing tumour.
A surprising finding is that when the tumour cells spread into the brain’s white matter, they actually become less aggressive. This is because the attempt of the tumour cells to repair the injured white matter causes them to mature into cells that resemble normal white matter brain cells, and in this mature state, the tumour cells are less able to grow and spread.
Using drugs that promote normal white matter repair, such as Pranlukast, currently an asthma drug, could be an effective way to treat glioblastoma, exploiting this natural mechanism to suppress tumour growth and spread.
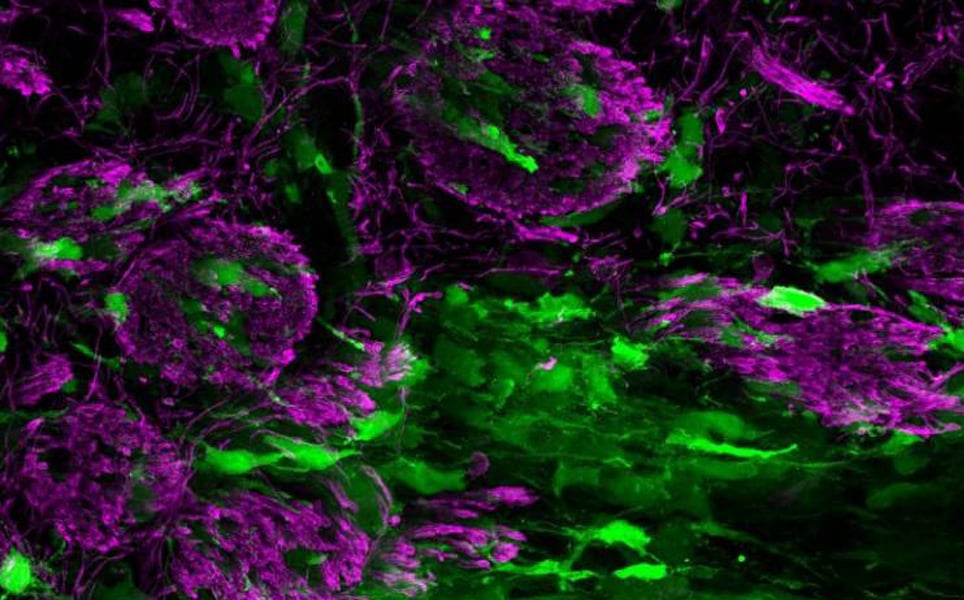
When glioblastomas become less aggressive
10. Step closer to nasal spray drug delivery for Parkinson’s disease
Significant progress has been made in the development of a nasal spray treatment for Parkinson’s disease by scientists at the University of York.
Levodopa is a drug that is currently being used to treat Parkinson’s disease. It is converted into dopamine in the brain, reducing the deficit of dopamine-producing cells in patients with Parkinson’s. After extended periods of use, the body starts to breakdown levodopa before it reaches the brain, and so increased doses are required. This means that instead of tablets, the drug needs to be injected. However, nasal sprays would be a much more effective way of delivering the drug straight to the brain.
To administer levodopa via a nasal spray, the drug needs to be able to adhere to nasal tissue long enough to release a sufficient dose. In this research, scientists developed a new gel that can adhere to nasal tissue alongside levodopa, enabling it to be released directly into the brain.
Results have so far shown that the gel enables levodopa to better adhere to tissue inside the nose, leading to increased levels of uptake into the blood and brain. The researchers are now working to progress the spray to human clinical trials.
Improved drug delivery
Banner image credit: Dr Owen Gwydion James, University of Edinburgh

)