
Scientifica's picks of the top neuroscience research from October 2021
Take a look at these ground-breaking neuroscience stories from October, including a visual prosthesis that enabled a blind person to see shapes, personalised on-demand DBS to treat depression, and detection of the earliest effects of Huntington’s in human embryos.
1. Our brains have a 'fingerprint' too
Scientists at EPFL have made the discovery that everyone has a unique brain fingerprint, which consists of signals of brain activity, and constantly changes over time.
Using MRI scans to produce graphs showing functional brain connectomes that indicate activity of neural networks, the researchers were able to see parts of the brain that are active when specific activities are being performed. When studying these brain connectomes, scientists at Yale University discovered that everyone has a unique brain fingerprint, with graphs generated from MRI scans of the same subject taken a few days apart being able to be correctly matched 95% of the time.
In previous studies, brain fingerprints were identified using MRI scans acquired over several minutes. However, researchers at EPFL discovered that they can be detected from around 1 minute 40 seconds of data. The study showed that the fastest brain fingerprints appear from sensory areas of the brain, especially those related to eye movement, visual perception and visual attention.
The researchers plan to compare the brain fingerprints of healthy patients compared to those suffering from Alzheimer’s disease, as initial findings suggest that the brain fingerprint features slowly vanish as the disease progresses.
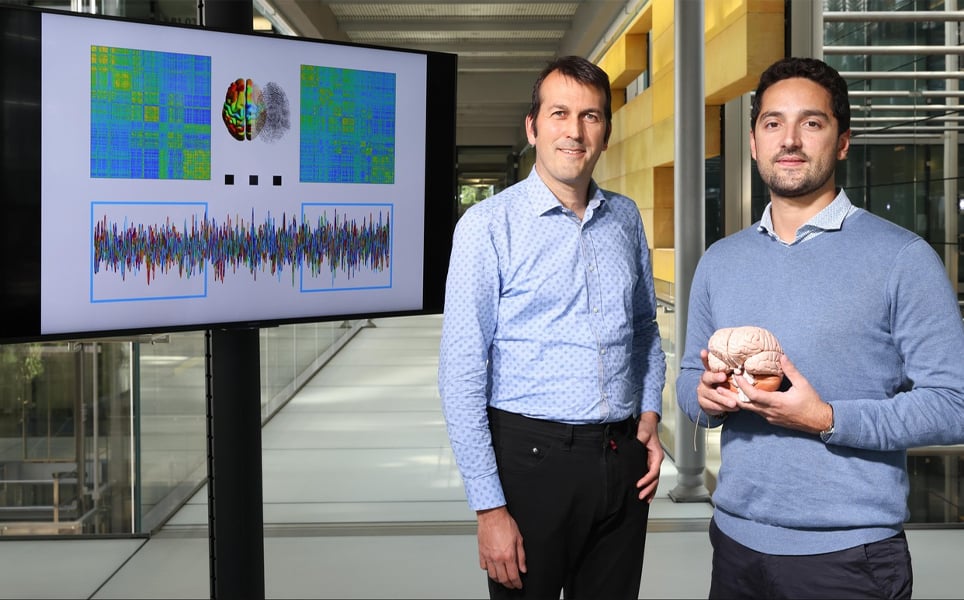
Identifying people from their brain fingerprint
2. Artificial networks learn to smell like the brain
Using machine learning, researchers at MIT have found that artificial neural networks can teach themselves to smell in just a few minutes, by mimicking the olfactory circuits in mammalian brains.
The algorithm used has no resemblance to evolution, yet seeing the artificial neural network so closely replicate biological structures suggests that the brain’s olfactory network is optimised for its function.
As it precisely matches the natural structure of circuits in the brain, the artificial network will be used as a model to understand more about the structure of the brain’s olfactory circuits, how they evolve under different conditions and how they can be manipulated.
Understanding how brain areas evolve for their function
3. Scientists enable blind person to see simple shapes using brain implant
By creating a form of artificial vision using a visual prosthesis, scientists at the John A. Moran Eye Center at the University of Utah and Miguel Hernández University have enabled a blind person to see lines and shapes.
A microelectrode array, which could both record and stimulate neuronal activity, was implanted into the visual cortex. The patient, Berna Gómez, wore glasses that contained a small video camera, which recorded visual data and sent this to the array via specialised software. The array then stimulated neurons to produce phosphenes, which were perceived by Gómez as white points of light, creating an image.
Gómez experienced no complications from the surgery, and the function of neurons was not impaired by the electrodes, demonstrating that the method is safe as well as effective. Just one array was placed in the brain, but other research suggests that placing between seven and ten arrays in the visual cortex could enable more detailed images to be produced in the brain of a blind person.
The aim of this and future research is to restore functional vision for people who have lost their vision, increasing their independence and safety.

Progressing prostheses to restore vision
4. Treating severe depression with on-demand brain stimulation
Researchers the University of California San Francisco have successfully treated a patient with severe depression by using a precision-medicine approach to modulate the specific brain circuit that is involved in causing depressive symptoms.
In this trial, the researchers discovered a specific pattern of brain activity that indicates the onset of symptoms, and therefore acts as a neural biomarker. A new deep brain stimulation (DBS) device was then customised to only deliver electrical stimulation to the brain when that pattern appears. Upon detecting the neural biomarker, the device stimulates the brain circuit, enabling individualised therapy to be delivered on-demand.
The patient’s symptoms were alleviated almost immediately, whereas standard treatment models take four to eight weeks. Symptoms were alleviated for the whole 15 months that the device was implanted in the patient’s brain.
The results of this study are promising for transforming the treatment of severe, difficult-to-treat cases of depression.
Improved DBS could transform treatment of depression
5. Study detects origins of Huntington's disease in two-week-old human embryos
Researchers at Rockefeller University have detected the earliest effects of Huntington’s in the first two weeks of human embryonic development. With symptoms typically emerging in midlife of adulthood, this finding is a breakthrough in understanding the causes and developing treatments for Huntington’s disease, with no cure or treatments currently available.
Huntington’s is caused by mutations in the Huntingtin gene, which lead to an unusually long protein being produced. Although the Huntingtin gene is expressed in every cell of the body, its functions are not known.
In this study, scientists created synthetic, lab-generated embryos, which are derived from stem cells and mimic the behaviour of human cells during development. Using the CRISPR/Cas9 gene-editing method, they inserted a range of mutations associated with Huntington’s into the embryos.
By comparing embryos with and without the mutations, they discovered that the mutations cause a visible change in the size of the embryonic germ layers. It is these germ layers that the progenitors of all brain cells emerge from. The researchers identified that this variation is caused by changes in the signalling pathway that guides embryonic cells.
It is not known exactly how these mutations affect development and how people with these mutations function normally for years, but it is suspected that a developing embryo may compensate for the effects of the mutations.
The researchers are now using the synthetic human embryos to screen for drugs that can correct these abnormalities.
Breakthrough in understanding Huntington's
6. Deterioration of brain cells in Parkinson’s disease is slowed by blocking the Bach1 protein, preclinical study shows
The Bach1 regulatory protein has been shown for the first time to have a role in in Parkinson’s disease by researchers at the Medical University of South Carolina (MUSC).
The research showed that Bach1 levels were increased in post-mortem brains of patients with Parkinson’s disease, and cells without Bach1 were protected from damage associated with Parkinson’s. Collaborating with vTv Therapeutics demonstrated that HPPE, an inhibitor of Bach1, protects cells from inflammation and oxidative stress when administered before or after the onset of disease symptoms.
Analysis of the entire genome of brains from Bach1-depleted mice showed that Bach1 represses gene expression of genes controlled by Nrf2, which switches on the expression of over 250 genes that protect cells from the stressors in Parkinson’s, as well as regulating expression of many other genes. The researchers concluded that a drug that inhibits Bach1 and activates Nrf2 would be most effective against Parkinson's.
Learn more
7. Psychologists create first ever body-maps of hallucinations
Body-maps of sensations arising during hallucinations in people experiencing psychosis have been created for the first time by scientists at Leicester University. This provides the most extensive descriptive data to-date on the feelings that arise during hallucinations and where sensations are felt.
The method involves creating multimodal unusual sensory experience (MUSE) maps by documenting hallucinations that occur in daily life, including the emotions experienced during hallucinations. The study discovered that hallucinations are characterised by multiple feelings arising at once, including multisensory, emotional and embodied features.
Results from this study will advance scientists’ understanding of hallucinations and how people who experience them can be supported.
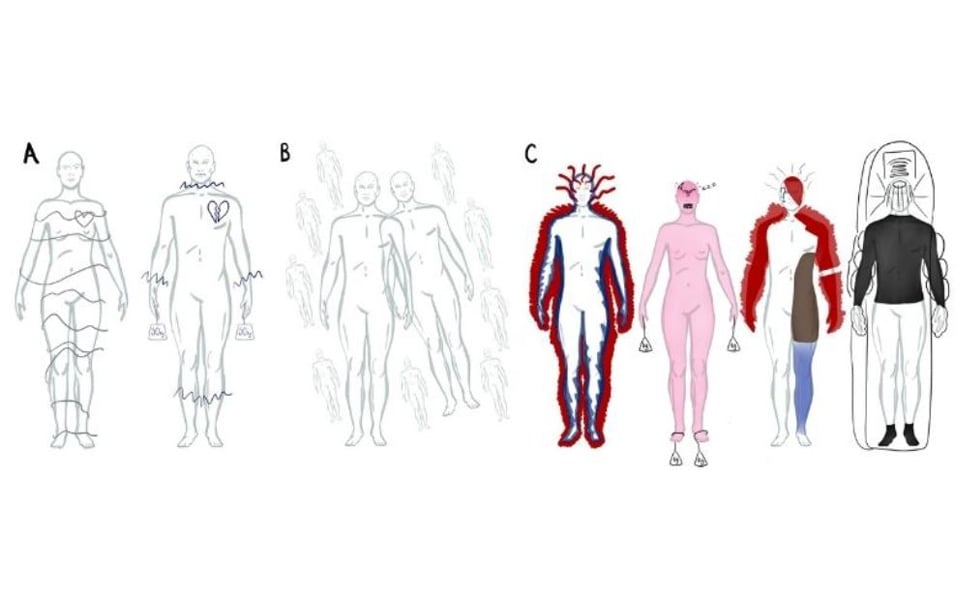
Mapping hallucinations
8. New initiative to improve sleep and waste clearance of the brain
A multidisciplinary team of researchers will be conducting the first human trial of a device that is designed to speed up and enhance the natural clearing of metabolic waste from the brain via the glymphatic system, that takes place as a person sleeps.
The trial is taking place at three sites: The University of North Carolina, University of Washington School of Medicine, and a collaboration between Oregon Health & Science University and the Brain Electrophysiology Laboratory (BEL). It is being funded by the Department of Defence, following recent studies identifying that the quality of sleep is important for clearance of brain metabolic waste through the glymphatic system.
Disrupted sleep means disrupted clearance of waste, leading to impaired motor coordination, attention deficits, slower processing speed, decreased decision-making abilities and impaired short-term memory, as well increased risk of neurodegenerative diseases in later life.
The technology being trialled is called Augmented Neural Oscillation Driver, ‘AugNOD’. AugNOD is an easy-to-use, wireless headband. It uses a combination of electroencephalography (EEG) and transcranial electrical stimulation, applying this before sleep to monitor and improve slow wave sleep and glymphatic clearance.
The study will also evaluate the validity of novel imaging and modelling approaches developed at the University of Washington School of Medicine that do not require contrast agents, which could improve the options for studying both short and long-term brain health.

Enhancing brain waste clearance
9. Scientists gain new understanding of how brain cells talk
Researchers at the University of Nottingham have increased our understanding of how brain cells perform synaptic communication, as well as identified a potential new molecular target for treating neurological and psychological conditions.
The study focused on synaptic RNA, which encodes proteins that are central to the function of nerve cells and synapses. It is thought that ‘tags’ label the RNAs to ensure they are located at the exact place where they are required for synaptic signalling, and some RNA has a methyl group added, which can influence proteins binding and stop proteins being produced.
The researchers found that RNA labelling can be reversed at synapses, and therefore may act as a synaptic tag. They discovered that methyl groups are removed when a synapse is active, and if this labelling process is disrupted, this can cause toxic protein clumps to form, affecting the function of nerve cells and synapses.
Targeting dysfunctions in this process could enable psychiatric conditions such as anxiety and addiction disorders, as well as early-stage neurodegenerative diseases to be reversed.
Insights into synaptic communication
10. Precision medicine data dive shows water pill may be viable to test as Alzheimer’s treatment
Analysis of patient data, mouse and human cell experiments and human population studies have been combined to discover that patients taking bumetanide, a commonly available oral diuretic approved by the FDA, have a significantly lower prevalence of Alzheimer’s disease compared to those not taking the drug.
The research team, led by scientists at Gladstone Institutes, the University of California, and the Icahn School of Medicine at Mount Sinai, is one of more than 20 teams supported by the NIA through a program encouraging researchers to use big data approaches to seek drugs that could potentially be repurposed.
They began by analysing data from 213 brain tissue samples, identifying expression of the APOE4 gene, which is a known genetic risk-factor for late-onset Alzheimer’s disease. They also analysed levels of gene expression of APOE4 carriers. Following this, the researchers compared the APOE4-specific signatures of Alzheimer’s against those of more than 1300 FDA-approved drugs, and from five drugs showing a gene expression signature that may help neutralise the disease, bumetanide was the strongest candidate.
To validate the data-driven discoveries, the scientists tested bumetanide in both mouse models of Alzheimer’s and induced pluripotent stem cell-derived human neurons, and found that treating mice expressing the human APOE4 gene reduced learning and memory deficits. Analysis of health records of adults over 65 taking bumetanide and those who did not, confirmed that those who had a genetic risk and took bumetanide had a ~35% to 75% lower prevalence of Alzheimer’s disease compared to those not taking the drug.
The results from this study are promising and provide evidence to justify a proof-of-concept trial of bumetanide as a treatment for those with a genetic risk of Alzheimer’s disease.
Using precision medicine to identify new treatment avenues
Banner image credit: istockphoto.com

)