
Know thy cells : Tips on how to patch cells in culture

by Guilherme Neves Virion, Post Doc, Burrone Lab at CDN, King’s College London
Introduction
I often hear people say that I'm lucky to work with cell cultures because they're so easy. But I can assure you, this is not the case. I have done electrophysiology experiments in both acutely dissociated and cultured cells as well as in brain slices (both freshly cut and cultured). All of these experimental systems have their own peculiar challenges, but one thing is true for all of them: healthy cells are a dream to patch, while sickly cells are a nightmare.
The good thing about working with cell cultures means you can separate the time when you maintain the optimal cellular conditions from the time when you do the actual recording. If you do your preparation right, you can come to the lab ready to record for a full day, and potentially record from 30 or more cells. The flip side, of course, is that if your cells are not right, there is nothing you can do at the patching rig to reverse the situation. You must go back to the drawing board.
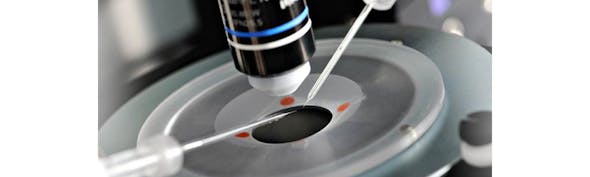
Here I share a checklist of factors that affect the success of patching in cultures. I will focus all the specifics on Human Embryonic Kidney 293 (HEK293) cell line, but these factors are true for any cells in culture. Before you start, make sure you research thoroughly how to keep your chosen cells as happy as possible (https://www.atcc.org/ is an excellent starting point). Also, make sure the cell line is suitable for patching. Cells that are very flat (I am talking to you Vero cells) will be very difficult to obtain Giga-seals. Check the literature for studies using your cells and “electrophysiology” or “patchclamp”.
If you can’t find any hits, there is probably a reason for it...
Density/passage number
This is the most basic determinant of cell health for most cell lines. Make sure you follow a defined protocol to keep density at optimal levels and passage numbers low. When it comes to preparing cells for patching, it is best to reduce density (to about 5K cells per cm2), to ensure cells are isolated. Ideally patch within 2 days of plating.
Adhesion
The easiest way to record from culture cells is to grow them in glass coverslips that you place on the tissue culture plastics and can quickly transfer to a recording chamber like one of these (https://www.warneronline.com/c...) under sterile conditions. As an aside these are also an excellent choice for immunocytochemistry applications. The downside is cells don’t like growing in glass as much as in tissue culture treated plastics. So, you will need to clean and coat the coverslips. We use Poly-L-Lysine, but collagen is also a good option for cancer derived cell lines.
Antibiotics
Removing antibiotics from the culture media will improve your success in patching cells. Ideally keep the cells for at least a week without antibiotics before patching. On a related note, regularly test for mycoplasma infections, as these will reduce your patching efficiency dramatically.
Transfection/siRNA treatments
These treatments can have a big impact in cell health. If you can work with stable clonal cell lines, your life will be greatly improved. These will help with reproducibility as well. If you must work with transiently transfected cells, make sure to wait as much as possible after the treatment, to allow time for cells to recover.
Perfusion and osmolarity
After taking care of your cells on the tissue culture incubator, make sure you also pamper them at the recording rig. Perfuse them with a HEPES based extracellular solution at a steady rate, keep them at a comfortable temperature and if they are showing signs of poor health (granules, swelling) slightly increasing the osmolarity of your solutions (both intra and extracellular) by 10-15% can help…
Summary
By following these tips, you can increase the chances of success when patching cells in culture.



)