
The most exciting neuroscience research from September 2021
A detailed cellular map of chronic lesions in MS, two promising new drugs for Parkinson’s disease, and an exciting discovery that could help with the development of treatments for addiction are included in Scientifica’s top neuroscience stories from September. We hope you find these interesting!
1. Algorithm can predict possible Alzheimer’s with nearly 100 percent accuracy
A deep-learning algorithm that can predict the possible onset of Alzheimer’s disease from fMRI images of the brain, with more than 99% accuracy, has been developed by researchers at Kaunas University.
Mild cognitive impairment (MCI) is often one of the first signs of Alzheimer’s disease, signifying the stage between the normal cognitive decline that happens with age, and dementia. Functional magnetic resonance imaging (fMRI) can be used to detect early stages of MCI, however identifying changes associated with Alzheimer's from fMRI images is time-consuming and requires specific knowledge.
In this study, the researchers found the deep-learning based method successfully analysed fMRI images obtained from 138 subjects, classifying them into six categories with an accuracy of 99.99%. This is promising data for the future of early Alzheimer’s disease diagnosis, which could improve treatment and outcomes for patients.
An algorithm to detect Alzheimer's
2. Scientists build a cellular blueprint of multiple sclerosis lesions
A detailed cellular map of chronic lesions in multiple sclerosis (MS) has been created by scientists at the National Institutes of Health’s National Institute of Neurological Disorders and Stroke (NINDS). This has enabled identification of genes responsible for lesion repair.
These chronic lesions, characterised by an inflamed, expanding rim of microglia immune cells, have been linked to more aggressive and disabling forms of MS. Microglia usually protect the brain, but in MS they can become overactive, secreting molecules that are toxic to nerve cells.
Using single-cell RNA sequencing to analyse gene activity, the researchers were able to create a comprehensive map of all cell types involved in chronic lesions, including their expression patterns and interactions. This revealed that, compared to normal tissue, the cell types in tissue surrounding chronic legions are much more diverse, with a high proportion of astrocytes being active at the edge of the lesions. The scientists also discovered that the gene for the complement component 1q (C1q), was mostly expressed by a subgroup of microglia, which were responsible for driving the inflammation. This suggests that this subgroup of microglia may play a crucial role in lesion progression.
Further studies in mouse models of MS confirmed the potential of C1q in human microglia as a new therapeutic target to treat chronic brain inflammation in MS.
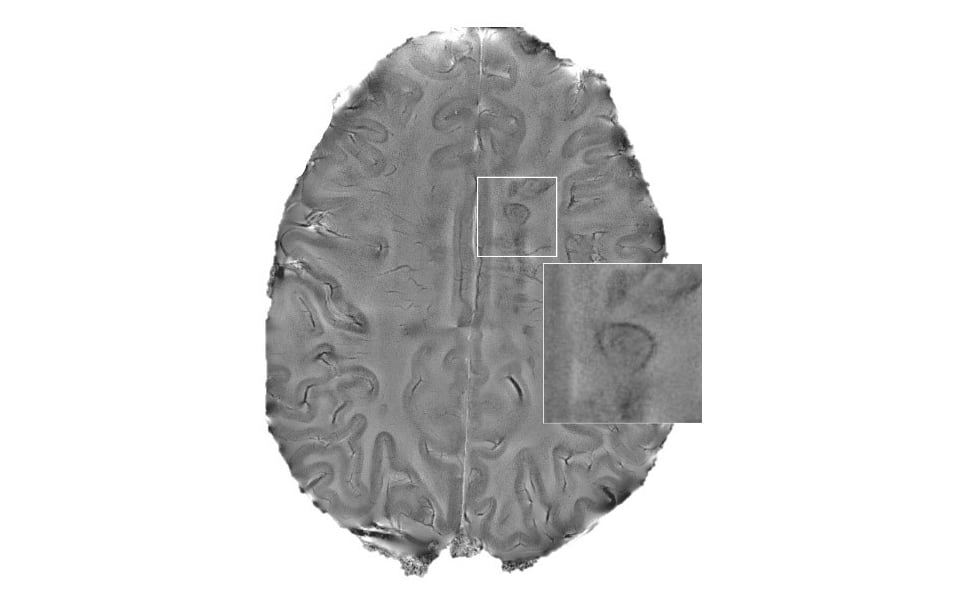
Cellular map to enhance understanding of MS
3. New gene therapy pathway could protect us from cancer and dementia
A new gene therapy that could protect against life-limiting diseases such as cancer and dementia has been discovered by scientists at the University of Sheffield.
Cancer, caused by uncontrolled cell proliferation due to genome damage, and neurodegeneration, caused by genome damage that leads to cell death, are both regulated by the newly discovered pathway.
The pathway involves three proteins, USP11, KEAP1, and SETX, which work together to repair DNA damage, to keep our DNA healthy and our cells functioning correctly. Understanding the role these proteins play in protecting cells from DNA damage will help make it possible for scientists to target these proteins to modify their behaviour, promoting genome and cell health and preventing diseases.

More about the newly discovered gene pathway
4. Research reveals drug targets for memory enhancement
Research led by the University of Bristol has identified new targets for memory enhancing drugs. Currently, there are limited treatments for memory loss. The discovery of new targets could lead to the development of treatments for a variety of brain disorders that involve memory loss, including Alzheimer’s disease and schizophrenia.
The researchers have identified specific receptors for acetylcholine, a neurotransmitter that is released during learning and is essential for the acquisition of new memories. These receptors have been found to redirect information that is transmitted through neural circuits in the hippocampus that encode memories.
Current treatments for symptoms of memory impairment in diseases such as Alzheimer’s target acetylcholine, but broadly boost levels of the neurotransmitter, which causes many adverse side effects. The discovery of specific receptor targets could enable development of drugs that generate the positive effects whilst minimising the negative ones.
Specific targets for memory enhancement
5. Nasal drugs show promise for slowing Parkinson’s disease progression
Two new nasal drugs, developed by Rush University Medical Center, have shown success in slowing down Parkinson’s disease progression in mice.
The drugs tested were two peptides, known as TLR2-interacting domain of Myd88 (TIDM) and NEMO-binding domain (NBD). Both were shown to slow inflammation in the brain and also slow the spread of the alpha-synuclein protein, which forms Lewy bodies in the brain of patients with Parkinson’s disease. Both treatments also improved the motor functions of the mice, including their gait and balance.
If the treatments also prove successful in humans, they could be a huge development in the treatment options for Parkinson’s, Lewy body dementia and other neurological disorders.
Peptides showing promise as Parkinson's treatments
6. New AI algorithm to improve brain stimulation devices to treat disease
An AI algorithm that can help elucidate which brain regions directly interact with each other has been developed by scientists at Mayo Clinic. This could help optimise electrode placement for stimulating devices that treat brain conditions such as Parkinson’s disease and epilepsy.
The algorithm, termed ‘basis profile curve identification’ makes it easier to determine how stimulating one area of the brain with brief pulses of electrical current affects other areas of the brain that directly interact with the stimulated region.
In the study, a patient with a brain tumour had an electrocorticographic electrode array placed in their brain to locate seizures and map brain function before removal of a tumour. This generated thousands of time points to study, with the algorithm making it easier to understand which regions were directly interacting.
A downloadable code package is available for other researchers to test the technique, with the hope of it leading to electrical stimulation being a viable treatment option for psychiatric illnesses and brain injuries.

Learn more
7. Withdrawal from psychostimulants restructures functional architecture of the brain
A team of researchers led by the University of California San Diego School of Medicine have made a potentially pivotal discovery in the pathway to developing treatments for addiction.
Mouse studies revealed how withdrawal from the addictive psychostimulants, nicotine, methamphetamine and cocaine, altered the functional architecture and patterns of brain activity. Each drug affected different regions of the brain, producing unique patterns of activity, yet withdrawal from the drugs has similar effects, with withdrawal from them all leading to a reduction in mortality.
When different brain regions interact with other regions performing similar functions, functional units known as modules are formed. Different modules control different processes, such as mood, motivation and sensation. In these studies, withdrawal led to a large decrease in the number of modules, as though the entire brain was processing the effect of lack of drugs. Functional connectivity throughout the brain also increased.
It is believed that the decreased mortality was a result of the restructuring of brain networks. The restructuring that occurs upon drug withdrawal from psychostimulants is likely why these drugs are so addictive, and could be a major target for treatments for addiction.

Possible new target for treating addiction
8. Docking peptides, slow to lock, open possible path to treat Alzheimer’s
Researchers at the University of Houston and Rice University have discovered that amyloid beta peptides and other small molecules in the brain go through several intermediate stages of frustration as they dock and lock onto the tips of growing fibrils in Alzheimer’s disease. This frustration during the growth of amyloid fibrils could be a potential treatment target.
Amyloid beta peptides seek the easiest path to bind to the tips of growling fibrils, but, when the positive and negative forces between atoms don’t align immediately, they are sometimes held back, also known as frustrated. Once the atoms align, the growing fibrils form the signature plaques of Alzheimer’s and other neurological diseases.
It is hoped that understanding this process could enable drugs to be developed that utilise the frustrated intermediate stages of peptide aggregation and stabilise the tips of fibrils, preventing further aggregation.
Preventing peptide aggregation
Banner image credit: Lauren Smith, UC San Diego Health Sciences

)