
Using cell-attached patch clamp to monitor neuronal activity
By Dr. Vincenzo Marra, University of Leicester
Here is a short introduction to the use of patch clamp electrophysiology in cell-attached configuration for monitoring neuronal activity. While a cell-attached configuration is often associated with the study of single channel currents, this method can be used to measure spontaneous firing activity, membrane potential and synaptic inputs without changing the cell's cytosolic content.
Find out more about neuronal electrophysiology here
When and why?
There may be a number of reasons to use cell-attached over whole-cell patch clamp to monitor neuronal activity, but the main one is perhaps that cell-attached does not modify the cytosolic content of a neuron. While whole-cell patch clamp allows you to measure currents across the entire neuron, when the patch pipette solution contacts the cytosol there will be an inevitable dilution of the intracellular content. After a few minutes in whole-cell configuration, the patch pipette solution will diffuse inside the patched neuron, changing the concentration of small signalling molecules (e.g. cAMP) and ionic species. It has been documented extensively that some electrical properties of neurons (calcium currents, firing rate etc.) change after several minutes of recording in whole-cell configuration. This phenomenon is generally called 'rundown' and has probably been observed by most careful patch-clampers.
In some cases, infusing a cell with a specific molecule may be desirable (e.g. intracellular blockers or fluorescent dyes) but it is also likely to affect the cell's spontaneous activity in ways that are hard to predict. Perforated patch clamp mitigates some of the cell dialysis issues associated with whole-cell but it comes with its own limitations and challenges. Cell-attached patch clamp is easy to implement and is one of the least invasive ways to monitor and manipulate the activity of a single cell. Here are a few considerations you may want to keep in mind when patching in cell-attached configuration.
Loose or tight?
Unlike whole-cell, the cell-attached configuration does not require reaching a giga-seal between the pipette and the cell membrane. If you are only interested in monitoring spike frequency or triggering action potentials, you may prefer a 'loose patch' with seal resistance in the order of MOhms rather than GigaOhms. Loose patch allows you to use the same pipette for multiple recordings and is particularly useful for identifying presynaptic neurons for paired recordings (see Bhumbra et al. 2018). However, a tight seal will allow you to record the membrane potential and synaptic inputs in current-clamp. As the seal resistance increases, the potential measured by your pipette will approach the neuron's membrane potential. For a full explanation see Perkins (2006), a must-read if you want to monitor neuronal activity using cell attached. Always measure your seal resistance several times during your recording; the seal may increase or decrease over the course of time, affecting your observed membrane potential.
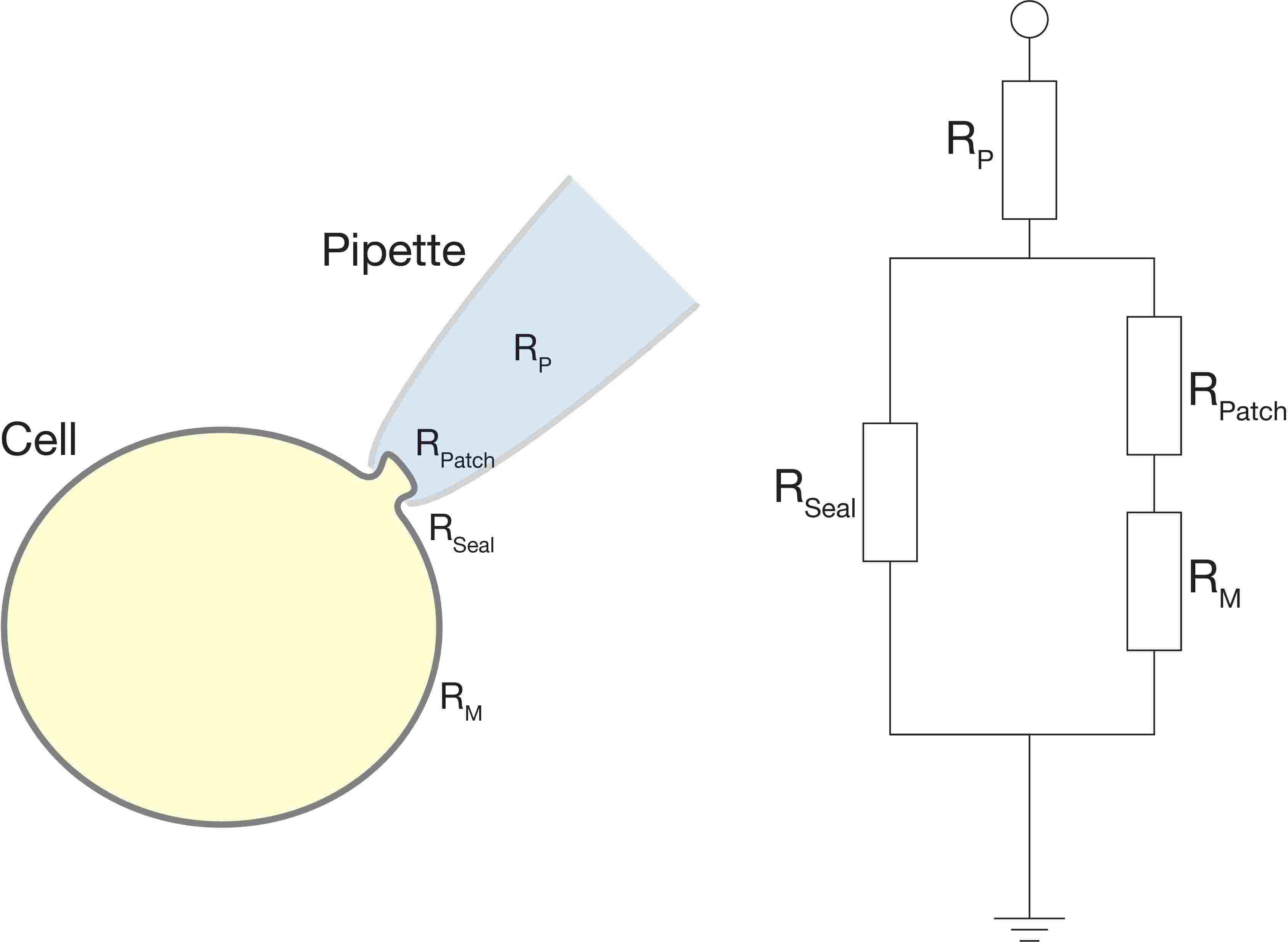
Figure 1: An electrical-equivalent circuit of a cell-attached patch clamp configuration. Read this article for more information on the electrical properties of a cell.
What is the potential across your patch?
A common mistake when performing cell-attached patch clamp for the first time is to forget that the pipette potential is applied outside the cell rather than inside. This is an easy mistake to make at the beginning, particularly considering that most modern amplifiers and software are labelled for whole-cell patch clamp. I find that the most intuitive way to correct this mistake is to think step-by-step about what is happening across a patch of membrane. When you reach a giga-seal holding the pipette potential at 0mV the patch of membrane will 'see' a more negative potential (~-70mV) on the intracellular face. However, if out of habit, you change your pipette potential to -70mV as you would before breaking-in for whole cell configuration, the patch of membrane will 'see' ~-70mV on the intracellular face and -70mV on the extracellular face. Effectively, the difference in potential between the inside and outside of the patch will be 0mV - a potential which is likely to activate most voltage-gated channels. Given the small amount of current required to hold the potential of a patch, the rest of the membrane will not be affected.
Another important consideration is that if your seal resistance is much higher than the patch resistance, the current generated to change the potential of your pipette will flow through your patch and through the rest of the cell's membrane, changing the neuron's membrane potential.
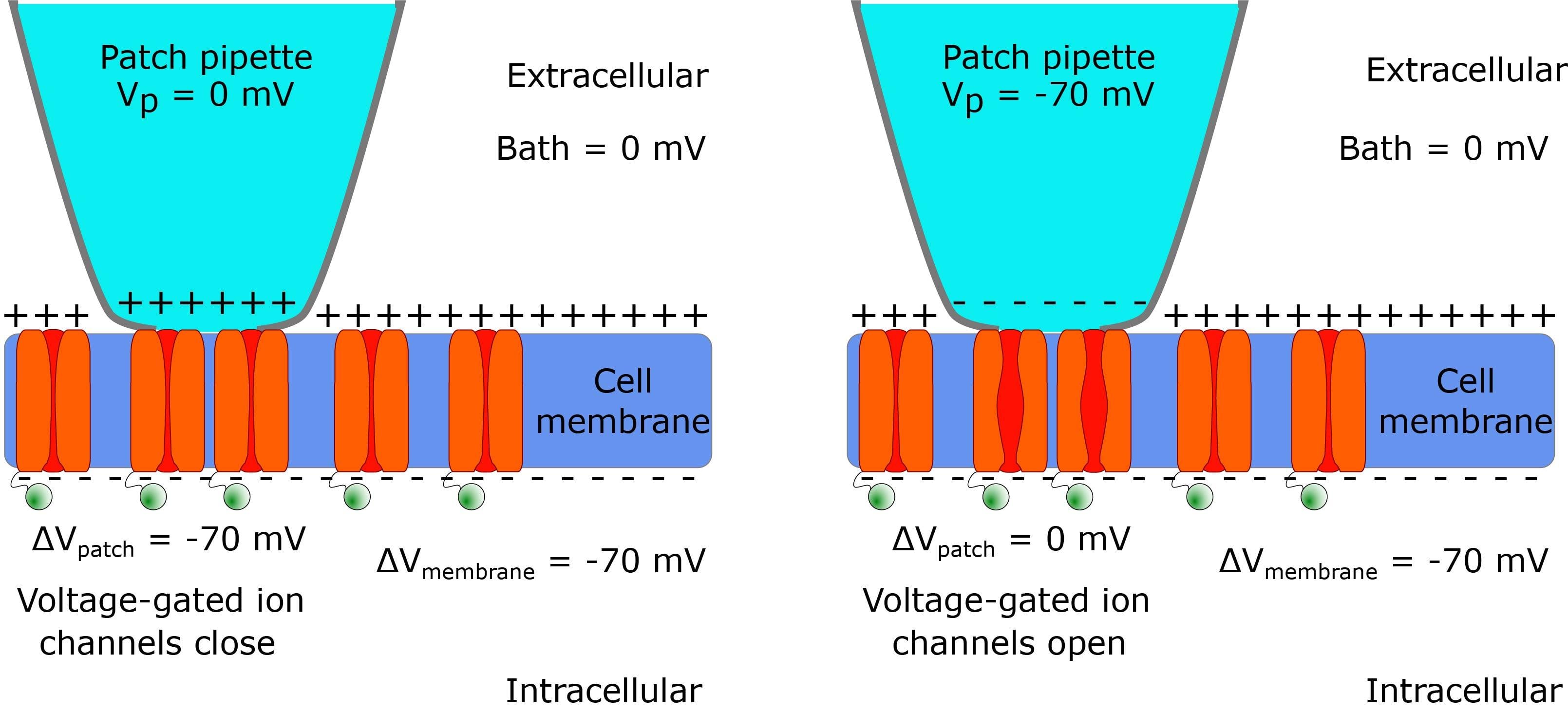
Figure 2: A diagram demonstrating that the pipette potential applied to the outside of the cell should be 0mV to prevent a change in the membrane potential.
Voltage-clamp or current clamp?
The choice between voltage-clamp or current-clamp will depend on the parameters you wish to measure, but in one sentence: use voltage-clamp to monitor and trigger action potentials and current-clamp to measure membrane potential or synaptic inputs. This may seem a little counterintuitive to patch clampers using voltage clamp to measure post-synaptic currents, but the principle is clearly explained again by Perkins (2006).
The pipette solution
Extracellular solution or artificial cerebrospinal fluid (aCSF) are common choices of pipette solution for cell-attached patch clamp recordings, particularly for loose patch. However, a simple solution with ~150mM sodium chloride is a good alternative with a very small junction potential. Solutions using bicarbonate/carbogen buffering are not ideal for long term recordings.
Read more about internal solutions here
References
1. Bhumbra, G.S., Beato, M., 2018. Recurrent excitation between motoneurones propagates across segments and is purely glutamatergic. PLoS Biol. 16, e2003586. https://doi.org/10.1371/journa...
2. Debanne, D., Guérineau, N.C., Gähwiler, B.H., Thompson, S.M., 1995. Physiology and pharmacology of unitary synaptic connections between pairs of cells in areas CA3 and CA1 of rat hippocampal slice cultures. J. Neurophysiol. 73, 1282–1294. https://doi.org/10.1152/jn.199...
3. Perkins, K.L., 2006. Cell-attached voltage-clamp and current-clamp recording and stimulation techniques in brain slices. J. Neurosci. Methods 154, 1–18. https://doi.org/10.1016/j.jneu...
4. Ogden, D, ed. 1994 Microelectrode techniques. The Plymouth Workshop Handbook. Cambridge, The Company of Biologists Ltd
Sign up to receive our latest news
Find out about Scientifica's latest product releases, company news, and developments through a range of news articles, customer interviews and product demonstration videos.



)